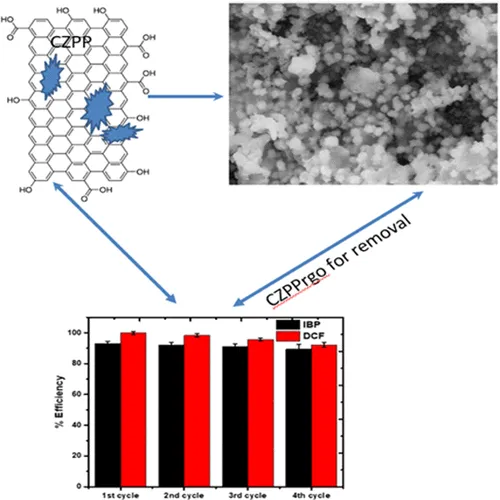
The persistent increase in the amount of nonsteroidal anti-inflammatory drugs such as ibuprofen (IBP) and diclofenac (DCF) in water bodies is alarming, thereby calling for a need to be addressed. To address this challenge, a bimetallic (copper and zinc) plantain-based adsorbent (CZPP) and reduced graphene oxide modified form (CZPPrgo) was prepared by facile synthesis for the removal of ibuprofen (IBP) and diclofenac (DCF) in water. Both the CZPP and CZPPrgo were characterized by different techniques such as Fourier transform infrared spectroscopy (FTIR), X-ray diffraction analysis (XRD), scanning electron microscopy (SEM), and pHpzc analysis. FTIR and XRD confirmed the successful synthesis of the CZPP and CZPPrgo. The adsorption of the contaminants was carried out in a batch system, and several operational variables were optimized. The adsorption is affected by the initial concentration of the pollutants (5–30 mg·L–1), the adsorbent dose (0.05–0.20 g), and pH (2.0–12.0). The CZPPrgo has the best performance with maximum adsorption capacities of 148 and 146 mg·g–1 for removing IBP and DCF from water, respectively. The experimental data were fitted into different kinetic and isotherm models; the removal of IBP and DCF follows the pseudo-second order, which can be best explained by the Freundlich isotherm model. The reuse efficiency was above 80% even after four adsorption cycles. This shows that the CZPPrgo is a promising adsorbent for removing IBP and DCF in water.